Testing and Evaluation of Orthopaedic and Spine Biomaterials
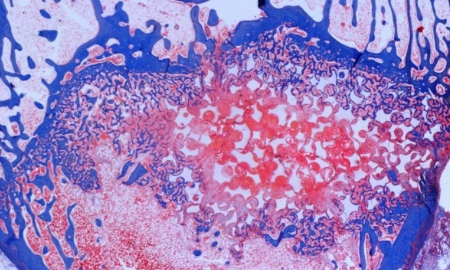
 The OREC Biomaterials and Histology Laboratory is actively involved in the testing and evaluation of orthopaedic and spine biomaterials in validated spine models.5 In addition to work on ceramic and metallic biomaterials for spine medical devices, BIMA has been involved in the evaluation of radiolucent spine fusion devices. BIMA has evaluated polylactides as resorbable spine devices.3,4,6 In addition, BIMA has published on the use of polyetheretherketone (PEEK) as a radiolucent non-absorbable spinal biomaterial.2 PEEK has well-established mechanical and good-wear characteristics, as well as excellent biocompatibility in both bulk and particulate form.1
The OREC Biomaterials and Histology Laboratory is actively involved in the testing and evaluation of orthopaedic and spine biomaterials in validated spine models.5 In addition to work on ceramic and metallic biomaterials for spine medical devices, BIMA has been involved in the evaluation of radiolucent spine fusion devices. BIMA has evaluated polylactides as resorbable spine devices.3,4,6 In addition, BIMA has published on the use of polyetheretherketone (PEEK) as a radiolucent non-absorbable spinal biomaterial.2 PEEK has well-established mechanical and good-wear characteristics, as well as excellent biocompatibility in both bulk and particulate form.1
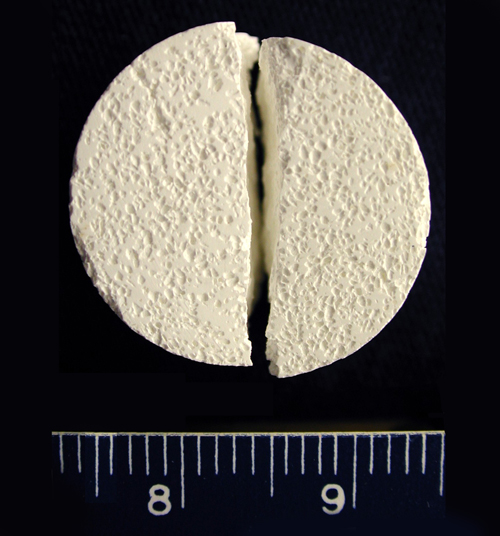
Image: Diametral Tensile Strength Test (DTS) test of a porous calcium phosphate cylindrical sample
Selected Publications
1) Toth JM: Chapter 8: “Biocompatibility of Polyaryletheretherketone Polymers.” In: The PEEK Biomaterials Handbook. 2nd Edition. Ed by Stephen M. Kurtz. Elsevier Publishing, Maryland Heights, MO, Pages 107-119, 2019.
2) Toth JM, Wang M, Estes BT, Scifert JL, Seim HB 3rd, Turner AS: Polyetheretherketone (PEEK) as a Biomaterial for Spinal Applications. Biomaterials 27(3): 324-334, January 2006.
3) Klopp LS, Toth JM, Welch WC, Rao, Tai JW, Thomas KA, and Turner AS: Bioresorbable Film for the Prevention of Adhesion to the Anterior Spine after Anterolateral Discectomy. Spine Journal, 9(5):411-417, May 2009.
4) Thomas KA, Toth JM, Crawford NR, Seim HB, and Turner AS: Bioresorbable Polylactide Interbody Implants In An Ovine Anterior Cervical Discectomy And Fusion Model: 3 Year Results. SPINE 33(7):734-742, April 1, 2008.
5) Easley NE, Wang M, McGrady L, and Toth JM: Biomechanical and Radiographic Evaluation of an Ovine Model for Human Lumbar Spine. Journal of Engineering in Medicine, Proceedings of the Institution of Mechanical Engineers, Part H, 222(6): 915-22, 2008.
6) Toth JM, Wang M, Scifert J, Cornwall GB, Estes BT, and Turner AS: Evaluation of 70/30 D,L-PLA for Use as a Resorbable Interbody Fusion Cage. Orthopedics, 25(10): S1131-1140, October 2002.
View more BIMA Research
Join BIMA
The Biomaterials & Histology Laboratory is looking for individuals with an interest in learning more about biomaterials or participating in collaborative research projects. For more information on becoming a member of BIMA, contact Dr. Toth.

 The OREC Biomaterials and Histology Laboratory is actively involved in the testing and evaluation of orthopaedic and spine biomaterials in validated spine models.
The OREC Biomaterials and Histology Laboratory is actively involved in the testing and evaluation of orthopaedic and spine biomaterials in validated spine models.