Contact Us
For additional information about the Computational Lung Physiology Laboratory, Contact the CLPL.
Report a Problem
To report a problem with this website, contact BME Communications or report an accessibility issue.
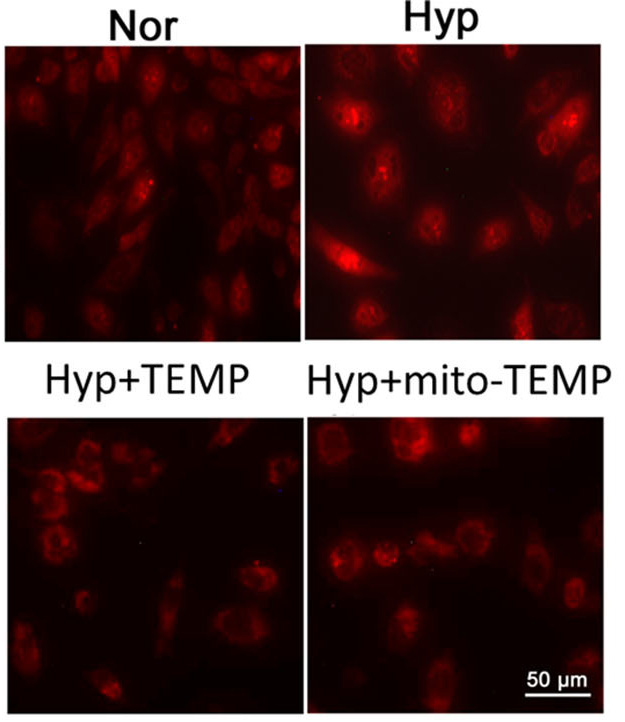 Nearly one million patients are placed on mechanical ventilation in ICUs annually. These patients often require high fractions of oxygen (hyperoxia) which damages and complicates the injury that triggered mechanical ventilation initially. Pulmonary endothelial cells lining the walls of the blood vessels in the lung are particularly sensitive to hyperoxia, exhibiting increased rates of production of mitochondrially-derived reactive oxygen species (mtROS), followed by increased microvascular permeability (e.g. pulmonary edema). The signaling mechanisms by which hyperoxia-evoked mtROS leads to pulmonary edema are not well understood. Thus, we are investigating the molecular basis of hyperoxia-associated mitochondrial-fragmentation in pulmonary endothelial cells and subsequent pulmonary microvascular permeability.
Nearly one million patients are placed on mechanical ventilation in ICUs annually. These patients often require high fractions of oxygen (hyperoxia) which damages and complicates the injury that triggered mechanical ventilation initially. Pulmonary endothelial cells lining the walls of the blood vessels in the lung are particularly sensitive to hyperoxia, exhibiting increased rates of production of mitochondrially-derived reactive oxygen species (mtROS), followed by increased microvascular permeability (e.g. pulmonary edema). The signaling mechanisms by which hyperoxia-evoked mtROS leads to pulmonary edema are not well understood. Thus, we are investigating the molecular basis of hyperoxia-associated mitochondrial-fragmentation in pulmonary endothelial cells and subsequent pulmonary microvascular permeability.
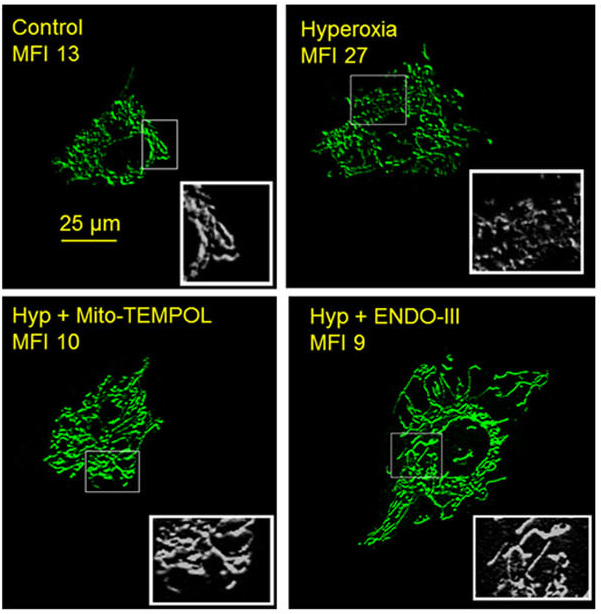 The first component of this work is to genetically manipulate fission/fusion proteins to determine the extent to which hyperoxia-induced mitochondrial DNA damage modifies expression/activation ratios of these proteins to enhance mitochondrial-fragmentation. The second is to demonstrate using both cellular and in vivo techniques that microvascular permeability leading to pulmonary edema is a consequence of mitochondrial DNA damage and augmented mitochondrial-fragmentation. Elucidating these mechanisms could lead to identification of therapeutic targets that could decrease morbidity in critically ill patients.
The first component of this work is to genetically manipulate fission/fusion proteins to determine the extent to which hyperoxia-induced mitochondrial DNA damage modifies expression/activation ratios of these proteins to enhance mitochondrial-fragmentation. The second is to demonstrate using both cellular and in vivo techniques that microvascular permeability leading to pulmonary edema is a consequence of mitochondrial DNA damage and augmented mitochondrial-fragmentation. Elucidating these mechanisms could lead to identification of therapeutic targets that could decrease morbidity in critically ill patients.
Hyperoxia Causes Mitochondrial Fragmentation in Pulmonary Endothelial Cells by Increasing Expression of Pro-Fission Proteins. (Ma C, Beyer AM, Durand M, Clough AV, Zhu D, Norwood Toro L, Terashvili M, Ebben JD, Hill RB, Audi SH, Medhora M, Jacobs ER) Arterioscler Thromb Vasc Biol 2018 03;38(3):622-635 PMID: 29419407
The Computational Lung Physiology Laboratory has a strong commitment to training the next generation of scientists interested in using state-of-the-art computational techniques to improve human health. Research opportunities are available for undergraduate and graduate (MS and PhD) students alike. For more information on joining our team, Contact the CLPL !